How Blood Extracellular Matrix Accelerates Early-Stage Healing in Animal Wounds
Discover how blood-derived extracellular matrix (ECM) regulates inflammation, supports angiogenesis, and accelerates early tissue repair in acute animal wounds
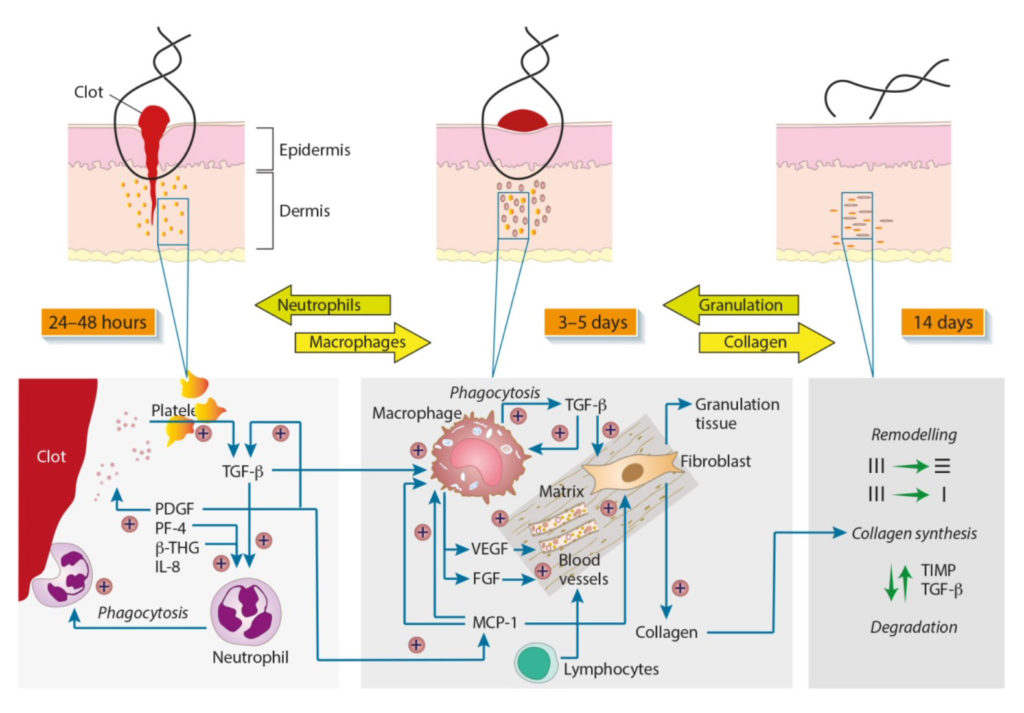
The blood extracellular matrix (ECM) plays a critical role in tissue repair, particularly during the early stages of wound healing, due to its biologically complex composition. In both life-preserving clinical veterinary settings and animal research models, this acellular network of proteins and signaling molecules coordinates cell recruitment, clot formation, regulation of inflammation, and the regeneration of damaged tissues.
Understanding how the blood ECM acts in acute wound repair not only offers new insights into healing biology but also hints at opportunities for therapeutic intervention to improve recovery both in animals and humans.
What Is the Blood Extracellular Matrix?
After an acute injury, the blood extracellular matrix creates a provisional matrix consisting of mainly fibrin, fibronectins collagens, proteoglycans and growth factors. Instead of merely being a structural “glue,” the blood ECM is able to modulate many critical cellular responses in healing.
The blood ECM is formed immediately upon bleeding and formation of a clot, which occurs after acute injuries such as lacerations, surgical incisions, or trauma in animals. The subendothelial matrix in blood vessels and tissues provides some of the adhesion sites for platelets, which start the clotting process and form a scaffold that halts bleeding and begins to attract cells.
Early-Stage Healing: Phases and the Role of ECM
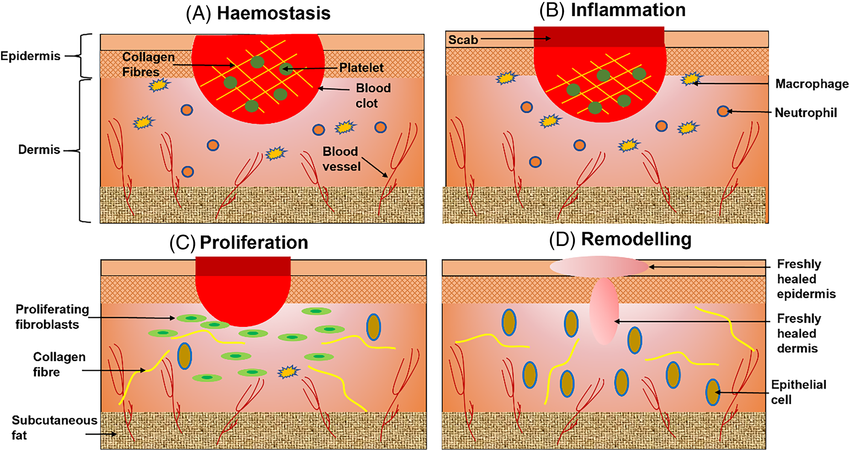
The healing of wounds proceeds in a tightly orchestrated but overlapping sequence:
- Hemostasis (minutes to hours)
- Inflammation (hours to days)
- Proliferation (days to weeks)
- Remodeling (weeks to months)
And within each phase, the blood extracellular matrix serves as both a structural support and biochemical mediator, hence orienting cells, modulating signaling, and steering the wound toward resolution.
1. Hemostasis: Quick Clot Formation Through ECM Interactions
From the instant a blood vessel is wounded, hemostasis must immediately start. When a vessel wall is damaged, platelets quickly adhere to the exposed proteins of the ECM (e.g., collagens). This initiates activation of clotting cascades, which results in a web of fibrin that constitutes the physical blood clot.
In this setting, the provisional blood extracellular matrix performs several critical roles:
- Arrests hemorrhage by securing an adherent clot.
- Preserves and concentrates growth factors in the clot matrix.
- Provides a door for the inflammatory and repair cells.
This acute matrix is replete with fibronectin, fibrin and collagen, all extracellular matrix (ECM) components that serve as binding sites for cells and counsel the early cell mediated responses.
2. Inflammation: Recruiting Cells and Signaling Repair
After clot formation, inflammatory cells such as neutrophils and macrophages are recruited to the wound by blood extracellular matrix signals. Integrins expressed on immune cells engage with ECM proteins, which attach and move into the wound.
EMC-associated macrophages additionally coordinate the switch from inflammation to proliferation through cytokine secretion including:
- PDGF (Platelet-Derived Growth Factor)
- TGF-β (Transforming Growth Factor-beta)
These cytokines induce cell migration, fibroblast activation and synthesis of new ECM components. This is important in animal wounds (as well as human ones), for regulated inflammation prevents too much tissue destruction and eases the way for repair, not long-term injury.
3. Proliferation: ECM Driven Tissue Formation
Cells including fibroblasts and endothelial cells move into the wound during the proliferation phase. The migration is directed by the blood extracellular matrix, which provides physical paths as well as chemical cues.
Key activities at this stage are:
- Granulation tissue, which is a dense network of blood vessels and ECM.
- Angiogenesis which is the process of new blood vessels buds under the guidance of ECM growth factors.
- Keratinocyte movement (reforming the protective layer of skin) on fibronectin and collagen gels.
Among the most abundant extracellular matrix (ECM) proteins in wounds is fibronectin, which has been of particular interest due to its role in providing a substratum for cell attachment and migration into the wound.
4. Remodeling: ECM Maturation and Wound Strength
At later phases of healing, the original blood extracellular matrix is replaced slowly by a fibroblast generated mature matrix. Type III collagen is initially deposited and eventually replaced by stronger type I collagen, thereby enhancing tensile strength.
The balance between ECM degradation and synthesis is modulated by MMPs and their inhibitors (TIMPs). Certainly, accurate regulation of ECM turnover is crucial on the part of animals, with disrupted ECM balance capable of preventing healing or causing overabundant scarring.
Why the Blood Extracellular Matrix Matters in Animal Wounds?
There are some of the biological responses in animal wounds (both domestic pet wounds or livestock wound) that parallel those of humans. Here’s why the blood extracellular matrix is particularly significant:
1. Provides Immediate Structural Support
Immediately or within a few minutes of the injury, it is blood ECM that orchestrates the wound signals. Without this temporary scaffold, cells are unable to move, and wounds do not advance well through the various stages of healing.
2. Signals and Modulates Cellular Behavior
It’s not like the ECM just “sits there”. It works actively with cells via integrin receptors and matricellular proteins, influencing:
- Fibroblast activation
- Immune cell polarization
- Endothelial cell behavior
These interactions determine how fast tissues regenerate and how effective they are in organizing newly formed tissue.
3. Guides Angiogenesis and Tissue Regeneration
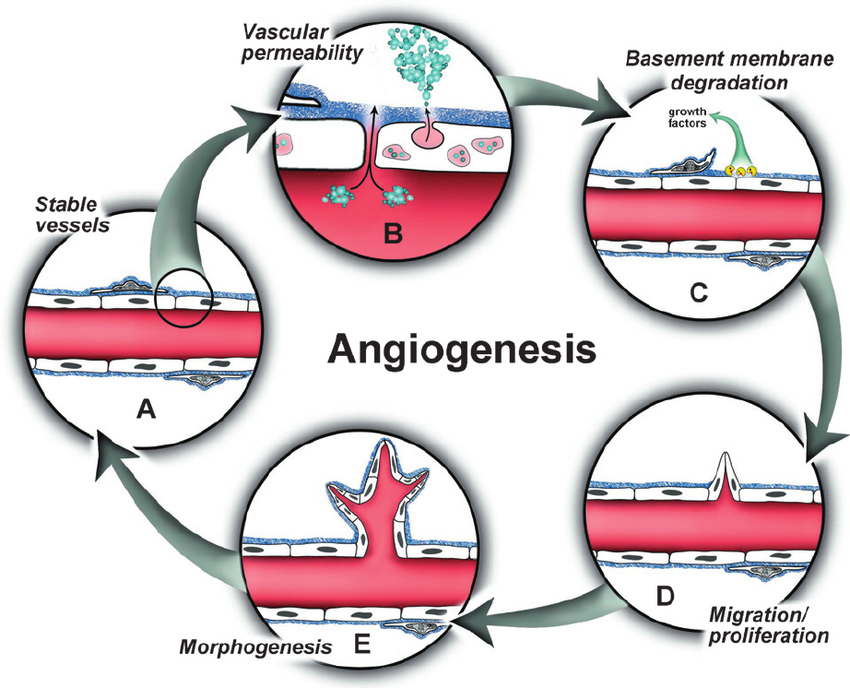
The formation of new blood vessels is necessary for supplying oxygen and nutrients to the tissue that is healing. VEGF and FGF are key angiogenic stimulators and, as ECM tethered growth factors, they regulate vessel formation within the wound bed.
4. Prevents Aberrant Healing and Scarring
Proper regulation of ECM turnover is paramount to avoid uncontrolled buildup of scar tissue. When there is increased or decreased deposition of the ECM, chronic wounds or fibrosis may occur and this can be especially problematic for animals with underlying conditions such as diabetes or infection.
Clinical and Veterinary Implications
1. Veterinary Wound Care
A greater knowledge of blood extracellular matrix may assist veterinarians in targeting therapies, such as ECM enhancers (dressings) or regulators of ECM turnover to enhance the healing process in animals.
2. Biomaterials and ECM Based Therapies
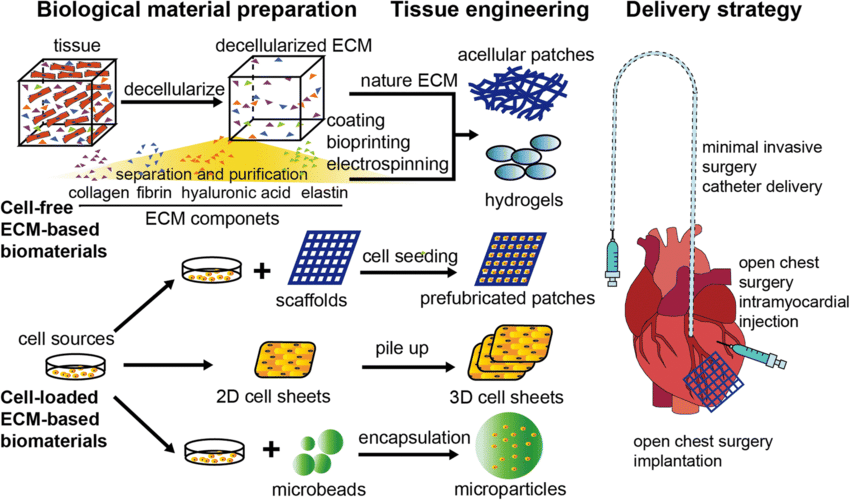
Current research in biomaterials seeks to create ECM mimicking scaffolds and formulations that aid healing, particularly in the case of challenging wounds for which conventional therapies are unsuccessful.
3. Future Research
Sectors like ECM based peptides and matrices have potential for enhancing wound management, minimizing scar formation and ultimately functional regeneration in animal as well as human patients.
Scientific Studies Supporting ECM’s Role in Wound Healing
Here are verified studies that validate the key roles of the blood extracellular matrix in wound healing:
Study 1: Wound Healing and ECM Dynamics
The extracellular matrix (ECM) is intimately associated with all phases of the wound healing process, including hemostasis to remodeling. This review described proteins such as collagen, elastin and glycoproteins that mediate cellular adhesion, migration and signaling crucial to healing.
Study 2: ECM Components Directly Regulate Fibroblast Function
Studies of dermal repair revealed that fibroblast survival, migration and metabolism regulated by ECM components are critical to the regeneration of connective tissue in acute wounds.
Study 3: MDPI Review on ECM Integration in Healing Phases
A comprehensive publication at MDPI demonstrated how the ECM regulates cell behaviour (e.g., adhesion and proliferation) in normal wound healing, and the importance of its synthesis throughout all phases.
Study 4: Cell Matrix Interactions in Dermal Repair
This review by BMC highlighted the direct contribution of ECM and cell interaction to the reparative process as well as in scarring and fibrosis regulation.
Conclusion
The blood Extracellular matrix is essential during the first stages of the acute wound healing process in animals. It’s not just structural scaffolding, it is an active regulator of cellular process, from clot stabilization and immune recruitment to regeneration and tissue remodeling.
The ability to appreciate and foster beneficial ECM activity may in the end serve to hasten healing, minimize morbidity, and enhance results in both veterinary and biomedical environments.


.png)



.webp)


.webp)
.png)